The AZAdvances Public Information Campaign shares stories of health solutions that are being discovered, developed, and delivered in Arizona. The initiative is led by AZBio in partnership with the Opportunity Through Entrepreneurship Foundation (OTEF). Working together, AZBio and OTEF are building AZAdvances to help Arizona healthcare innovators move these innovations forward faster.
Learn more at AZAdvances.org

Addressing the "Who Knew" Effect.
Arizona life science innovators are working to discover, develop, and deliver life saving and life changing innovations.
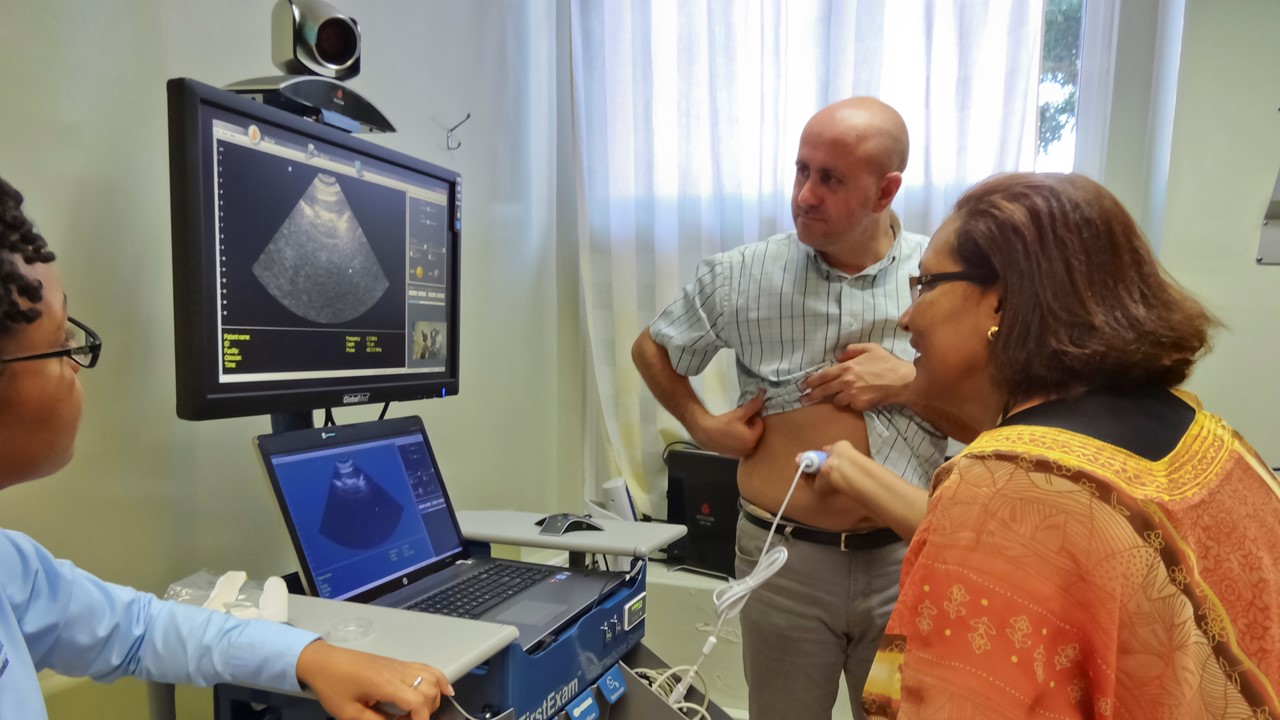
AZBio is pleased to partner with ABC15 and CW61 to share quick Sonoran Living Health Innovation Spotlights to help our community learn more about the work of Arizona's healthcare innovators.
Each segment touches on a public health challenge or opportunity and spotlights an Arizona life science company's work to create a solution.
You can view all spotlights below.
Sonoran Living Health Innovation Spotlights and Sonoran Living segments are paid for by AZBio.
Innovation Quick Links
AZAdvances
AZAdvances is an initiative created with support from AZBio to advance health innovation in Arizona within the Opportunity Through Entrepreneurship Foundation (OTEF).
AZAdvances programs focus on
- supporting the advancement of Arizona discoveries into development;
- commercialization path ways so discoveries can become treatments and cures;
- providing students with opportunities for connections, internships, and scholarships so that they can explore careers in this exciting field and we can create a long-term talent base in Arizona.
AZAdvances leverages the experience and expertise of leading organizations, decades of data, and a 3-year best practices study. The creation of AZAdvances by OTEF and AZBio is thanks to support from a grant from the U.S. Economic Development Administration, an agency in the United States Department of Commerce and a growing community of AZAdvances supporters.
Join us in supporting AZAdvances. Donate Here.
Brain Tumors
It is estimated that 700,000 Americans are living with a primary brain tumor and that an estimated 88,970 people will receive a primary brain tumor diagnosis this year.
According to the National Brain Tumor Society, often, low-grade tumors (grade I and II), which are not aggressive, are treated with watchful monitoring or surgery alone. Though all tumors are monitored with repeat scans, grade II tumors are watched more closely after surgery and over time to make sure there is no recurrence.
Higher grade tumors (grade III and IV), which are malignant and can grow quickly, are more difficult to remove and require additional treatments beyond surgery, such as radiation, chemotherapy, or a clinical trial if one is available. Microscopic tumor cells can remain after surgery and will eventually grow back. All treatments, therefore, are intended to prolong and improve life for as long as possible.
Driven to overcome the limitations of current treatments for brain tumors and raise the standard of care, a team of brain tumor specialists joined forces and formed GT Medical Technologies, Inc., to improve the lives of patients with brain tumors. GammaTile Therapy received FDA 510(k) regulatory clearance for the treatment of all types of recurrent brain tumors and newly diagnosed malignant tumors. 3D, resorbable collagen tiles embedded with radiation sources are implanted during the last five minutes of brain tumor resection, providing an immediate, dose-intense treatment to eliminate residual tumor cells. This "one and done" procedure allows patients to receive their course of radiation while going about their daily lives, requiring no additional trips to the hospital or clinic for radiation therapy. GammaTile Therapy is available in top brain tumor centers across the United States.
Vaccines
People in Arizona are rolling up their sleeves and getting a shot in the arm. COVID-19 vaccines are a hot topic in Arizona. How do we know that they are safe? Would you encourage someone you love to get one?
Questions about vaccines are natural and people need to have access to FACTS. A great science based resource is covidvaccinefacts.org
Vaccines are an important part of our health tool kit. Not just for COVID but for a wide range of preventable diseases. Now is the time to be sure that all of us are up-to-date on ALL of our vaccinations.
CDC recommended vaccination schedules for children and adults
Vaccine Innovation
Innovators in Arizona are hard at work exploring how we can create new vaccines for a wide range of diseases. One example is Calviri, led by Dr. Stephen Johnston with technology developed at the ASU Biodesign Institute. Calviri is developing a cancer vaccine. They are starting with dogs and if successful, people may be next.
Calviri’s mission is to provide affordable products worldwide that will end deaths from cancer by combining a unique, highly immunogenic source of neoantigens with a peptide array platform. On our course to eradicate cancer we will develop new diagnostic and therapeutic products, and then preventative vaccines that will stop cancer from starting.

Help Your Heart
Your heart is arguably the hardest working muscle in your body. With every beat, your heart pumps out about 70 milliliters (two ounces) of blood every time it beats. Over the average person’s lifetime, it will beat over three billion times. Unfortunately, more than 23 million people worldwide suffer from heart failure. It is the number one cause of medical problems and death in the developed world.
When you choose healthy behaviors, you can lower your heart disease risk while also preventing other serious chronic conditions like type 2 diabetes and even some kinds of cancer. The CDC recommends 7 Tips for a heart healthy lifestyle, check them out here.
Healing Hearts
Heart Disease Can Damage Your Heart Muscle.
Avery Therapeutics, Inc. is an Arizona company dedicated to developing tissue engineered therapeutics to treat diseases and injuries to human muscle. Avery’s lead product, MyCardia™, is being developed to address and treat heart failure. MyCardia is a stable and robust allogeneic engineered tissue graft that can be manufactured at scale, cryopreserved, and shipped at 4C. In pre-clinical studies of heart failure, MyCardia resulted in improved left ventricular volume (systole and diastole) and myocardial mass, in addition to improved quality of life metrics such as daily activity and exercise tolerance, compared to untreated cohorts. Note: MyCardia is in development and is not currently available for patients.
Learn more about Avery Therapeutics at averythera.com
Heart Disease
According to the CDC, Heart disease is the #1 cause of death worldwide for both men and women. The term “heart disease” refers to several types of heart conditions. The most common type of heart disease in the United States is coronary artery disease (CAD), which affects the blood flow to the heart. Decreased blood flow can cause a heart attack.
Learn more about Heart Disease at CDC.gov and at the American Heart Association.
When we think about heart disease, we often think of heart attacks, but there are many forms of heart disease and many causes. One of the most challenging is when both sides of the heart are no longer able to pump enough of blood. This is called end-stage Bi-Ventricular Heart Failure. When this occurs, the patient may need a heart transplant. But, they may not be strong enough for the surgery OR a donor heart may not be available in time.
SynCardia temporary Total Artificial Heart
SynCardia Systems in Tucson is the sole manufacturer of the only FDA-approved total artificial heart. This FDA cleared device is currently used as a bridge to transplant. The device has two main components, the TAH which is inside the patient and an external pump. As the blood flow improves, the patient can get stronger and be better prepared for when a donor heart becomes available. Patients from 8 to 80 have received a total artificial heart. The team at SynCardia recently celebrated the 2,000th patient that received this life saving technology.
SynCardia was founded in 2001 by renowned cardiothoracic surgeon Jack G. Copeland, MD; biomedical engineer Richard G. Smith, MSEE, CCE; and interventional cardiologist Marvin J. Slepian, MD. In clinical use for more than 35 years, the SynCardia temporary Total Artificial Heart (TAH) is the most widely used and extensively studied total artificial heart in the world.
To learn more about SynCardia
TRIF is a Smart Investment
Valley Fever
Innovation at Arizona's Public Universities
For 20 years, every person who pays sales tax in Arizona has been investing in life saving and life changing research efforts and may not even know it. The Technology and Research Initiative Fund or TRIF for short was created in 2000 as part of Prop. 301 which added six tenths of 1 percent to the state sales tax to fund K-12 education, workforce development and a small fraction for research at our three state universities.
Those fractions added up. Thanks to TRIF, Arizona’s universities are home to world class researchers who work to explore new ways to keep us healthy and help us when we are sick. AND – during the COVID-19 pandemic, have answered the call to help us all by expanding our testing capacity, providing important health data, and more. Today, our universities are collaborating to find better solutions for Arizonans living with Valley Fever.
According to a study by the Milken Institute, commissioned by the Flinn Foundation,
“the continued existence of this fund, and the commitment to research and technology it represents, is an essential component of Arizona’s innovation economy.”
Twenty years of TRIF supported research has helped to build up Arizona’s research infrastructure and our talent base. TRIF researchers explore big questions that impact our daily lives.
Learn more about TRIF
Learn more about Valley Fever

Health Innovations for Babies & Children
When it comes to treatments and cures, babies and children need and deserve a special innovation focus.
One example is INC - the International Neonate Consortium led by the Critical Path Institute in Tucson. Launched on May 19, 2015, INC is C-Path’s ninth consortium – a global collaboration formed to forge a predictable regulatory path for evaluating the safety and effectiveness of therapies for neonates. “By uniting stakeholders from research institutions, drug developers, regulatory agencies, patient advocacy and other organizations,” said Janet Woodcock, Director of the U.S. Food and Drug Administration’s Center for Drug Evaluation and Research (FDA/CDER), “INC can develop practical tools that can be incorporated into clinical trials for neonates, which will then lead to more successful, efficient trials and provide this population with better treatments.”
Learn more about the International Neonatal Consortium (INC)
Shining a light on babies
Innovations in Neonatal Jaundice
Neonatal Jaundice is a common and usually harmless condition in newborn babies that causes yellowing of the skin and the whites of the eyes. In mild cases this usually goes away after 1 or 2 weeks. But sometimes a baby needs more help in the form of a medical treatment called phototherapy or light therapy.
The team at Neolight, a medical device company in Scottsdale Arizona, created Skylife to help babies with jaundice. Unlike larger systems that can only be used in a clinical or hospital setting and often result in parents and their baby being separated during the precious early days of life, Skylife is small, portable, and can even be used in a home setting allowing Parents and Baby the opportunity to stay together and bond.
Learn more about Neolight
Developing Cancer Cures
Developing Cancer Cures
The more we learn about Cancer, the more complicated it gets. There are more than 100 different types of cancer. Plus, each patient is unique and additional genomic changes can occur as cancer cells evolve. When you factor all this in, the number of possible combinations is larger than the number of stars in the universe.
The talented team at Systems Oncology in Scottsdale is using Artificial Intelligence or AI for short to translate this massively complex scientific data into useful biological insights and novel cancer therapies with unprecedented speed, scale, and precision.
Learn more about Industry Investments in Cancer Drug Development
Systems Oncology, Inc.
Systems Oncology is developing a new class of multimodal RNA therapeutics that can simultaneously silence numerous disease targets with a single RNA molecule to improve outcomes and combat emerging drug resistance.
The company's Arromer platform is used to digitally engineer and rapidly produce self-delivering therapeutics which target specific tissues with unprecedented precision and accuracy. Systems Oncology is focused on building a pipeline of products in oncology, yet their multimodal RNA platform can be harnessed to transform outcomes for any complex polygenic disease.
Learn more about Systems Oncology
Harnessing the power of the immune system
Immunotherapy is a treatment that works by boosting the immune system in order for our body to fight cancer better, but it does not work for every patient. Based on the Phoenix Biomedical Campus and using technology licensed from ASU, OncoMyx Therapeutics has discovered a way to use a virus that infects rabbits but does NOT hurt people to make immune therapies even more effective and is working to develop new medicines and combinations of medicines that can save more lives.
Learn more about OncoMyx Therapeutics
Colon Cancer Awareness
March is Colon Cancer Awareness Month but being proactive and health aware is important every month.
Colorectal cancer is the third most common cancer in the US, and the second leading cause of cancer death. It affects men and women of all racial and ethnic groups, and is most often found in people 50 years or older. However incidence in those younger than 50 is on the rise. This disease takes the lives of more than 50,000 people every year. Colorectal cancer is also a highly preventable disease with screening.
Learn more about Colorectal Cancer
Saving Lives with Colorectal Cancer Screening
Colorectal Cancer screening is now easier than ever before. From at home screening tests to blood tests, colon cancer screenings save lives. Here in Arizona, Beacon Biomedical's laboratory developed test (LDT) solutions help primary care providers screen their patients for the presence of undiagnosed cancer to improve their chances of survivability. BeScreened™–CRC , offers a simple blood-based solution to patients who are unwilling or unable to use traditional colorectal cancer screening methods. In 2016, Beacon Biomedical received the AZBio Fast Lane Award for their work in developing the BeScreened-CRC test.
Learn more about Beacon Biomedical
Sun Safety & Skin Cancer Awareness
May is Skin Cancer Awareness Month. Here in Arizona protecting your skin is important every day of the year.
According to the University of Arizona Cancer, skin cancer is the most common form of cancer in the United States. It is estimated that more than 3.5 million new cases are diagnosed each year — more than all other cancers combined. One in five Americans will get skin cancer in their lifetimes. Skin cancers can affect your appearance as well as your health. Protecting your skin with sunscreen, wearing hats and protective clothing, and staying out of the sun at peak times is sun smart.
You also need to be vigilant. The best way to find skin cancer, before it becomes a serious problem, is by checking your skin regularly. Most of the brownish spots on your skin – freckles, moles, and birthmarks – are normal, but some may be skin cancers. It is important to look for changes in these spots or the appearance of new spots when checking your skin.
Learn more about how to be sun safe!
In the second video, listen to how experts in the field of dermatology utilize the DecisionDx-Melanoma test to better characterize individual risk of recurrence and metastasis in their melanoma patients.
Learn more about Castle Biosciences, Inc.
Telemedicine
Arizonans have been pioneering in telemedicine for decades. At our universities, in our innovative companies, and across our healthcare systems Arizona is home to leaders in using telehealth tools to make life better. These technologies are making healthcare more accessible and often more affordable too.
GlobalMed is a virtual health technology solutions leader that empowers providers to connect with patients and deliver the health services and medical expertise they need —whenever, wherever. Secure technologies support evidence-based care across specialties and acuity levels, driving efficient and expanded access to healthcare. With over 40 million teleconsults delivered in nearly 60 countries and specializing in both federal and commercial spaces, their advanced telehealth solutions are used worldwide from the VA and DoD to rural American hospitals and villages in Africa. Founded in 2002 by a Marine Corps Reserve Veteran still serving as CEO.
Learn more about GlobalMed
Pain Management
Pain is our body’s way of telling us that something is wrong or needs attention. Sometimes it may make us uncomfortable, for some people it can be debilitating. Pain medicines designed to address high pain levels, like opioids, serve a purpose in specific cases. They also can lead to severe or life-threatening consequences when not used correctly.
Learn more about the CDC's Rx Awareness campaign
New options to treat acute and chronic pain.
Regenesis Biomedical, an Arizona-based medical device company, has developed Provant Therapy which treats the pain site using pulsed electromagnetic energy that creates an energy field set to specific parameters, shown to reduce pain and inflammation. This FDA cleared device is providing doctors and patients with new options to treat acute and chronic pain.
Learn more about Regenesis Biomedical
Innovations in Food Safety
A case of food poisoning can really spoil your day. The CDC estimates 48 million people get sick, 128,000 are hospitalized, and 3,000 die from foodborne diseases each year in the United States. In 2021, over 11.5 MILLION pounds of food was recalled by the FDA from just 7 suppliers and there were more food recalls. With teams in Scottsdale and Tucson, PathogenDx has developed a rapid food environmental monitoring test to help food producers identify food-borne pathogens in the same shift to test their processes and products before they ship to help keep your food free of the contaminants that can make you sick and so that they can avoid the need for recalls.
Learn more about PathogenDx.
UArizona Agricultural Resources Span the State
The UArizona Cooperative Extension takes the science of the University to the people of Arizona through programs, publications, classes, events and one-on-one teaching.
Take a look at their areas of expertise and many locations
Important Information for Patients
This website and this web page are designed to spotlight Arizona-based healthcare innovations and innovators. The Arizona Bioindustry Association, Inc. (AZBio), our AZAdvances team, and our partners are not providing medical advice. If you have a personal health related question, contact your healthcare professional.