SynCardia Secures $19M Financing for Total Artificial Heart
$15M Financed by Athyrium Opportunities Fund, $4M by Existing Shareholders
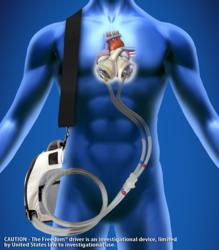
The Freedom® portable driver is the world’s first wearable power supply for the SynCardia temporary Total Artificial Heart. The Freedom driver has supported more than 120 patients worldwide.
Tucson, Ariz. (PRWEB) March 20, 2013
SynCardia Systems, Inc., the privately-held manufacturer of the world’s first and only FDA, Health Canada and CE (Europe) approved Total Artificial Heart, announced today the raise of $19 million of long-term growth capital, including a $15 million structured financing from Athyrium Opportunities Fund, as well as a $4 million follow-on equity investment from existing shareholders.
“The proceeds will help support commercialization of our Freedom® portable driver*, the world’s first wearable power supply for the SynCardia temporary Total Artificial Heart, and accelerate the launch of our new, smaller 50cc Total Artificial Heart**, designed for patients of smaller stature, including women and adolescents,” said Michael Garippa, SynCardia Chairman/CEO/President. “In 2012, we generated $25 million in revenue and a record-breaking 125 implants at more than 50 SynCardia Certified Centers worldwide.”
“SynCardia’s recent growth is likely the beginning of a longer-term trend,” said Laurent Hermouet, a Partner of Athyrium. “The Total Artificial Heart will soon be able to address the entire biventricular heart failure market thanks to newer offerings like the Freedom driver as well as the 50cc version of the Total Artificial Heart. This broader product offering coupled with convincing clinical and INTERMACS data makes partnering with SynCardia a compelling opportunity. We look forward to expanding this initial relationship as might be needed in upcoming quarters.”
SynCardia is an innovative, 85-employee company focused on advanced medical technology targeting the NYHA Class IV heart failure market. The company certifies and supports the top transplant centers around the world, including Texas Heart Institute in Houston, Cedars-Sinai in Los Angeles, Cincinnati Children’s Hospital Medical Center, La Pitié Hospital in Paris and the Heart & Diabetes Center NRW in Bad Oeynhausen, Germany. There are currently 80 SynCardia Certified Centers worldwide with an additional 32 hospitals undergoing the company’s four-phase certification program.
Athyrium Opportunities Fund (“Athyrium”) is a New York-based fund focusing on investment opportunities in the global healthcare sector. The Athyrium investment team has substantial healthcare investment experience across a wide range of asset classes, including public equity, private equity, fixed income, royalties, and other structured securities. Athyrium invests across healthcare verticals, including biopharma, medical devices and products, and healthcare services. The team partners with management teams to implement creative financing solutions to companies’ capital needs. For more information, please visit http://www.athyrium.com.
*CAUTION – The Freedom portable driver is an investigational device, limited by United States law to investigational use.
**In January, the U.S. Food and Drug Administration (FDA) approved two Humanitarian Use Device (HUD) designations for SynCardia’s smaller 50cc Total Artificial Heart to be used for destination therapy and pediatric bridge to transplant. The next step is for SynCardia to submit a Humanitarian Device Exemption application for each indication to the FDA for approval. Once approved, the HDEs will allow up to 4,000 U.S. patients annually to receive the 50cc Total Artificial Heart as destination therapy, and an additional 4,000 pediatric patients to receive the device as a bridge to transplant.
###
About the SynCardia temporary Total Artificial Heart
SynCardia Systems, Inc. (Tucson, AZ) is the privately-held manufacturer of the world’s first and only FDA, Health Canada and CE approved Total Artificial Heart. Originally used as a permanent replacement heart, the SynCardia Total Artificial Heart is currently approved as a bridge to transplant for people suffering from end-stage heart failure affecting both sides of the heart (biventricular failure). There have been more than 1,100 implants of the Total Artificial Heart, accounting for more than 300 patient years of life.
Similar to a heart transplant, the SynCardia Total Artificial Heart replaces both failing heart ventricles and the four heart valves, eliminating the symptoms and source of end-stage biventricular failure. Unlike a donor heart, the Total Artificial Heart is immediately available at SynCardia Certified Centers. It is the only device that provides immediate, safe blood flow of up to 9.5 liters per minute through each ventricle. This high volume of safe blood flow helps speed the recovery of vital organs, helping make the patient a better transplant candidate.
Forbes Ranks SynCardia #69 Among “America’s Most Promising Companies” In its February 2013 issue, Forbes selected SynCardia as one of “America’s Most Promising Companies” for the second consecutive year. On the list of 100 privately held, high-growth companies with bright futures, SynCardia was selected #69, moving up eight spots from its #77 ranking last year. See the full list of SynCardia Awards & Recognition here.
Contact
Don Isaacs SynCardia Systems, Inc. (520) 955-0660 Email
